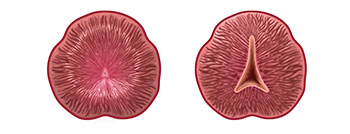
Views of the urethra before (left) and after (right) enlarged prostate (BPH) treatment with the iTind, a temporarily implanted nitinol device that reshapes the urethral prostate by gently expanding over 5 days to create channels through which urine can flow. A new study shows that the iTind provides rapid and effective relief of enlarged prostate symptoms.
CENTER VALLEY, Pa., (February 4, 2021) – Olympus, a global technology leader in designing and delivering innovative solutions for medical and surgical procedures, announced today the publication of a US multicenter study showing that the iTind provides rapid and effective relief of symptoms through a catheter-free procedure that preserves sexual function for men with an enlarged prostate. Titled, The iTind Temporarily Implanted Nitinol Device for the Treatment of Lower Urinary Tract Symptoms Secondary to Benign Prostatic Hyperplasia: A Multicenter, Randomized, Controlled Trial, the study was published December 25, 2020 in Urology®, a peer-reviewed medical journal known as the “Gold Journal.”
“This study demonstrates the safety and effectiveness of the iTind, a temporarily implanted device, in reshaping the prostate and allowing urine to flow, thus relieving lower urinary tract symptoms without the need for patients to go home with a catheter,” said lead author Dr. Bilal Chughtai, Associate Professor of Urology at Weill Cornell Medicine. “With such a common condition, it is important to offer treatments that are straight forward to perform and have the least side effects. Most adverse events experienced by patients during the trial were mild and resolved within two weeks, which is a major advantage over other therapies. Additionally, trial results showed that treatment with the iTind does not compromise sexual function.”
In this multicenter, randomized, controlled trial, 185 men aged 50 years or older with BPH (Benign Prostatic Hyperplasia), also known as enlarged prostate, were randomized 2:1 between iTind and sham procedure arms. Patients were assessed at baseline, one and half months, three months, and 12 months post-operatively for IPSS, peak urinary flow rate (PFR), residual urine, quality of life (QoL), and the International Index of Erectile Function (IIEF). A total of 118 received the iTind versus 57 receiving the sham treatment. At 12 months, the iTind group reported a reduction of 9.25 points in IPSS (p<0.0001), a 3.52ml/s increase in PFR (p<0.0001) and a 1.9-point reduction in QoL (p<0.0001). Most adverse events were mild and expected, such as dysuria (22.9%) and light hematuria (13.6%), and were self-resolving within 1-4 weeks.1 No de novo ejaculatory or erectile dysfunction occurred.
BPH is one of the most common diseases in aging men and the most common cause of LUTS (lower urinary tract symptoms), which 8 out of 10 men will face in their lifetimes.2,3 According to study findings, treatment with the iTind brings about rapid and sustained improvement in LUTS while preserving sexual function, a key concern for men considering BPH treatment options. Treatment with the iTind device avoids complications associated with drugs, surgery, or permanent implants, and it allows men to go back to their normal lives faster than they would be able to with other treatment options.
How Treatment with the iTind Works
- The iTind device is placed in the prostatic urethra during a procedure that can be done in a medical office setting or other site of care.
- Made up of three flexible nitinol (nickel titanium super alloy) struts, the iTind gently expands and reshapes the prostate by creating channels through which urine can flow. This process occurs over a five- to seven-day period and does not require a hospital stay.
- The device is then removed and the patient experiences immediate relief.
- Since no heat or permanent implant is used with the iTind procedure, there is typically no need to leave a Foley catheter in the urethra following the procedure.
- Leaving no permanent implant, the iTind procedure does not hinder future treatment options.
The iTind device was developed by the Israeli-based medical device manufacturer Medi-Tate. Through an investment in Medi-Tate, Olympus holds the exclusive right to distribute Medi-Tate products in the U.S., including the iTind device.
“We are thrilled to now have the U.S. based trial results available that further prove the efficacy of the iTind device for treating the symptoms of enlarged prostate,” said Ido Kilemnik, Founder and CEO of Medi-Tate, manufacturer of the iTind device. “The body of evidence is now robust with both U.S. and European studies having been published, and we look forward to making this device more widely available.”
“The iTind study demonstrates that patients treated with iTind in a real-world setting consistently experience relief from their symptoms with no long-term side effects. We are enthusiastic about the study results because they support broader adoption of this novel, effective BPH treatment,” said Randy Clark, President of the Medical Systems Group at Olympus America Inc. “The study results support use of this minimally invasive BPH treatment as an alternative to medications, which can have unpleasant side effects. The iTind provides many men with a non-surgical option beyond medication and permanent implants.”
# # #
About the COPD Foundation
Olympus is passionate about the solutions it creates for the medical, life sciences, and industrial equipment industries.
Olympus’ Medical business uses innovative capabilities in medical technology, therapeutic intervention, and precision manufacturing to help healthcare professionals deliver diagnostic, therapeutic, and minimally invasive procedures to improve clinical outcomes, reduce overall costs, and enhance the quality of life for patients. Olympus’ Medical portfolio includes endoscopes, laparoscopes, and video imaging systems, as well as surgical energy devices, system integration solutions, medical services, and a wide range of endotherapy instruments. For more information, visit https://medical.olympusamerica.com.
About Medi-Tate
Medi-Tate is an Israeli medical device company that deals in the R&D, manufacture and sale of innovative solutions for the treatment of Lower Urinary Tract Symptoms (LUTS) with a mission to commercialize a safe and effective, office-based solution for BPH.
1 Chughtai B, Elterman D, Shore N, et al. The iTind Temporarily Implanted Nitinol Device for the Treatment of Lower Urinary Tract Symptoms Secondary to Benign Prostatic Hyperplasia: A Multicenter, Randomized, Controlled Trial [published online ahead of print, 2020 Dec 26]. Urology. 2020;S0090-4295(20)31520-X. DOI: 10.1016/j.urology.2020.12.022
2 Medical Student Curriculum: Benign Prostatic Hyperplasia (BPH). Auanet.org. https://www.auanet.org/education/auauniversity/medical-student-education/medical-student-curriculum/bph. Published May 2013, Updated July 2016. Accessed January 18, 2021.
3 Lim KB. Epidemiology of clinical benign prostatic hyperplasia. Asian J Urol. 2017;4(3), 148–151. doi:10.1016/j.ajur.2017.06.004

